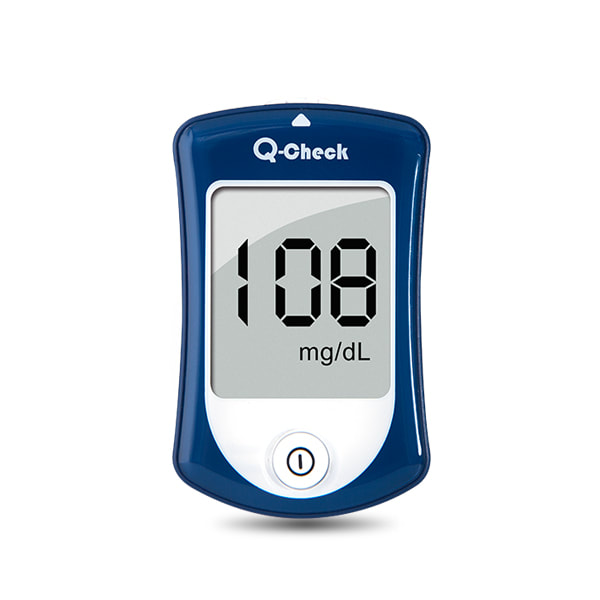
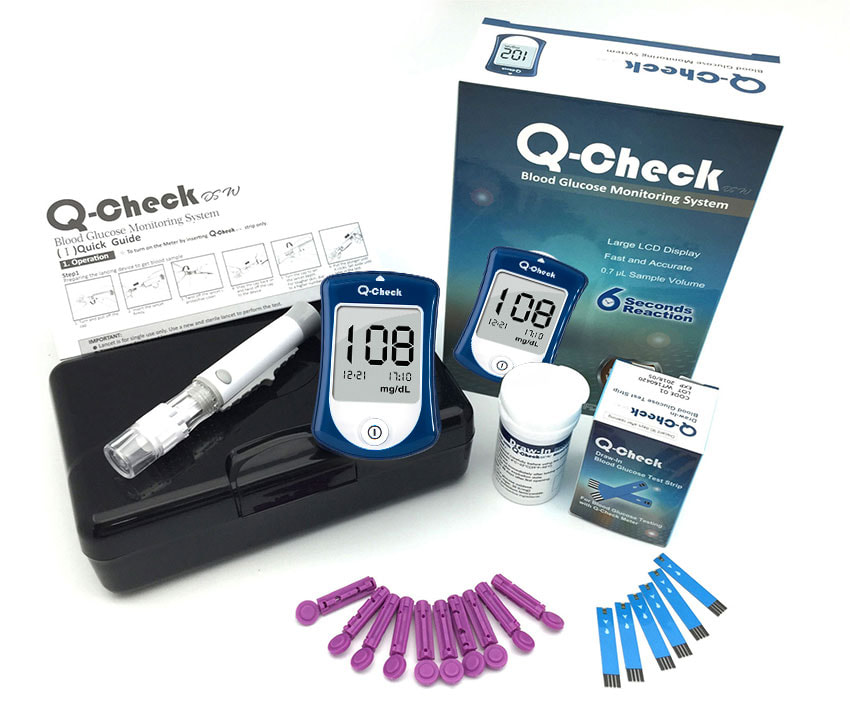
Blood Glucose Meter KitQ-Check
Anti-Bacteria surface and coding-free design, easy for user manage the diabetes.
With CE, FDA, ISO and GMP standard. |
Introduction
FDA CERTIFIED Q-CHECK BLOOD GLUCOSE MONITORING SYSTEM
Q-check Blood Glucose Monitoring System is FDA cleared, and meets GMP, ISO 13485 and CE mark standard.
Performing a glucose test with Q-check, you have different options of testing parts on your body besides the fingertip like palms or forearms as an alternative.
Q-check Blood Glucose Monitoring System is FDA cleared, and meets GMP, ISO 13485 and CE mark standard.
Performing a glucose test with Q-check, you have different options of testing parts on your body besides the fingertip like palms or forearms as an alternative.
Feature
- 0.7 µL sample volume
- 6-Second reaction time
- 500 memories with date and time
- 7-, 14-, 30-day average
- Ketone warning
- No coding
- Alternative site testing
- Anti-Bacteria surface
- Texas instruments chip inside
Specification
| Assay Range | 20 to 600 mg / dl (11.1 to 33.3 mmol/L) |
|---|---|
| Accuracy | 90% in zone A of EGA |
| Sample Volume | 0.7µL |
| Reaction Time | 6 seconds |
| Operational Temperature | 10 to 40°C ( 50 to 104°F) |
| Relative Humidity | 10 to 90% |
| Hematocrit | 20 to 60% |
| Battery Type | CR2032 3V lithium coin battery |
| Dimension | 84 x 55 x 13 mm |
| Weight | 42.8g without battery |
| Certificate | FDA, GMP, CE0123 |